Happy New Year!
Journal Club December:
JAAD:
Original article
Diffuse variants of scalp lichen planopilaris: Clinical, trichoscopic, and histopathologic features of 40 patients
Michela Starace, Gloria Orlando, Aurora Alessandrini, Carlotta Baraldi, Francesca Bruni, Bianca Maria Piraccini. Volume 83, ISSUE 6, P1659-1667
What: Retrospective study examining 40 patients affected by diffuse hair thinning associated with a long-lasting history of itching and erythema of the scalp and a histopathological diagnosis of LPP.
Why: To better describe Fibrosing Alopecia in a Pattern Distribution (FAPD) and Cicatricial Pattern Hair loss (CPHL) as variants of LPP.
Methods: Epidemiological; clinical (subjective symptoms and area of scalp involvement); trichoscopic features; treatment outcome; and long-term follow-up data were collected from outpatient clinical reviews, between 4/15-4/18.
Exclusion: incomplete clinical data; diagnosis other than LPP.
Demographic: all caucasian. M:F = 15:25. Mean age 54.9 (34-76 range).
Results: 18 diagnosed with FADP, 2 with CPHL, and 20 with a new variant which the authors describe as “Lichen Plano-Pilaris Diffuse Pattern” (LPPDP).
Clinical: FADP – mean age 54.9, mostly post-menopausal women; moderate pruritus/pain and had visible erythema; 4 of which has coexisting FFA based on clinical features; pull test pos in 83.3% (n=15); often misdiagnosed as AGA
CPHL – ages 72 and 76, mostly post-menopausal women; had previous mild pruritus but asymptomatic at assessment; no visible erythema but had focal atrichia; central scalp; pull test pos in 0%; often misdiagnosed as AGA
LPPDP – mean age 53, no sex predeliction; all had diffuse moderate/severe pruritus and visible erythema in 65% (n=13); diffuse scalp involvement pull test pos in 95% (n=19); often misdiagnosed as seborrheic dermatitis
Trichoscopic: FADP – all had loss of follicular ostea + white fibrotic patches; perifollicular erythema + follicular hyperkeratosis; no tufted hair
CPHL – both had loss of follicular ostea + white fibrotic patches; no perifollicular erythema + no follicular hyperkeratosis; no tufted hair
LPPDP – all had loss of follicular ostea + white fibrotic patches; perifollicular erythema + follicular hyperkeratosis in 70% (n=14) + tufted hair in 45% (n=9) + broken hair in 20% (n=4)
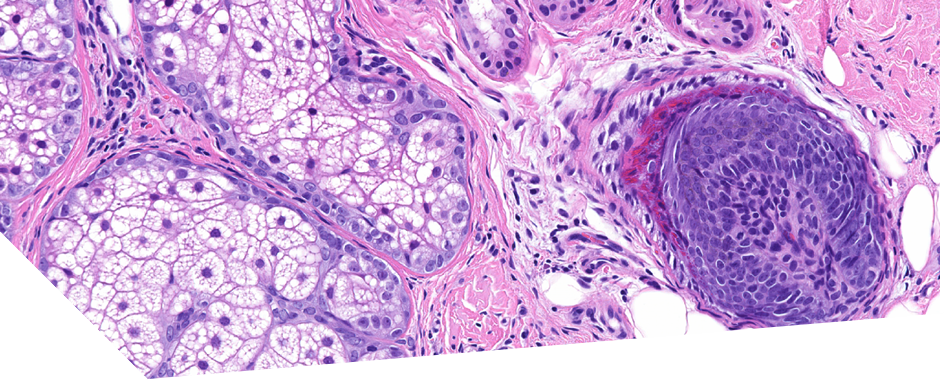
Histo: FADP/CPHL – reduced hair follicle number; decreased/absent sebaceous glands; mild lichenoid infiltrate around the isthmus and infundibular region and perifollicular lamellar fibrosis that mainly affected miniaturized follicles; spares terminal hairs. FADP had more prominent interface dermatitis whereas CPHL had prominent fibrotic collagen tracts
LPPDP – similar to above except the infiltrate and lamellar fibrosis spared miniaturised follicles and mostly affected terminal and intermediate follicles; prominent lymphocytic interface dermatitis
Treatment and outcome:
FADP – 9 had finasteride/dutasteride; 4 had hydroxychloroquine; 5 received IM triamcinolone; all also received topical treatment with either dermovate/tacrolimus/pimecrolimus and also topical 2 or 5% minoxidil solution; disease progression arrested in all.
CPHL – both received finasteride + topical 0.1% tacrolimus ointment + topical 5% minoxidil solution; disease progression arrested in all.
LPPDP – 14 had longer courses of IM triamcinolone cf FADP/CPHL; 8 had hydroxychloroquine; 15 had dermovate in addition to systemic; all had 2 or 5% minoxidil solution; disease progression arrested in 95% (n=19)
Author conclusions: 3 variants of LPP have been described in the study which show diffuse alopecia as opposed to classical LPP which has defined patches of scarring alopecia
FADP and CPHL may be more consistent with FFA, whereas LPPDP is more consistent with classical LPP
LPPDP is a new variant which has not been described before
CPHL might represent a post-inflammatory variant of FADP
Consider lichenoid alopecias in those with long-standing scalp itch/pain and diffuse hair loss
Limitations: small sample size
Thoughts: some would argue that CPHL is end-stage AGA with follicular drop-out and fibrosis and is not an LPP variant. It is possible that many patients with FADP and CPHL have coexisting AGA or that AGA triggers or at least contributes to both their pathogenesis.
Research letter:
The effect of intradermal botulinum toxin on androgenetic alopecia and its possible mechanism
Uri Shon, Myung Hwa Kim, Dong Yoon Lee, Se Hwan Kim, Byung Cheol Park. Volume 83, ISSUE 6, P1838-1839.
What: Prospective study
Why: DHT induces TGFB1 in dermal papilla cells (DPC’s), suppressing follicular epithelial cell growth. Antagonism of TFG-B1 may therefore be a target for treatment in AGA. Botulinum toxin type A (BTX) may inhibit TGF-B1 in DPC’s as has recently been used to treat AGA.
Methods: enrolled according to basic and specific classification. BTX injection as below. TGF-B1 expression from cultured DPC’s, exposed to 10-9mol/L DHT was evaluated by reverse PCR. Affect of BTX on DHT induced suppression of DPC TGF-B1 analysed via IMF.
Demographic: 18 males, mean age 49
Spectrum: baseline severity not stated
Exclusion- those treated with finasteride, minoxidil or supplements that affect hair growth
Exposure- Intradermal BTX every 4 weeks for 24 weeks. During each session, 30 units of BTS injected at 20 different sites. Hair regrowth after BTX analysed via unblinded phototrichogram image analysis at weeks 0, 12 and 24
Results: Mean hairs/cm2 +/- SD was: 129.61 ± 28.05 at week 0; 129.11 ± 28.80 at week 12; and 136.22 ± 33.05 at week 24
Hair number significantly increased at week 24 (P = 0.012) but not at week 12 (P = 0.803)
DHT upregulated the TGF-β1 expression of DPCs in 96 hours, whereas BTX downregulated the TGF-B1 expression in 96 hours
No adverse events
Author Conclusions: BTX successfully abrogated DHT-induced secretion of TGF-β1 from DPC
Intradermal injection of BTX was effective against AGA by inhibiting TGF-β1 secretion in the hair bulb
Thoughts: is the response robust, need long term follow-up
Until this was published the mechanism was felt to be due to relaxation of muscles increasing follicular oxygenation.
Research Letter
Improvement of 11 patients with nail psoriasis with apremilast: Results of an investigator-initiated open-label study
Allen S.W. Oak, Hoang Ho-Pham, Boni E. Elewski. Volume 83, ISSUE 6, P1830-1832
What: open-label, single- arm study analysing effect of apremilast on psoriatic nail disease
Why: Apremilast has previously improved NAPSI scores in ESTEEM 1 and ESTEEM 2 trials, measured as a secondary end-point. Limited licensed treatment options for nail psoriasis specifically.
Methods:
Demographic: 11 otherwise well adults, F:M = 5:6, mean age 47.7
Spectrum/inclusion: mean PASI 4.5, >1 fingernail involved + modified NAPSI (mNAPSI) >=5 + nail pain visual analogue scale >=4. mNAPSI scores range from 0 (no nail disease) to 130 (complete nail involvement in all nails).
Exclusion: those on phototherapy, systemics or other topical agents
Exposure: Apremilast 30mg BD for 52 weeks
Outcomes: primary end-point = mean percentage change mNAPSI score at 36 weeks cf baseline of all nails
Secondary end-points - mean percentage change in mNAPSI of the target nail (nail with highest baseline mNAPSI) at weeks 12, 24, 36, 48, and 52
Proportions of patients achieving an mNAPSI ≥75% reduction over baseline mNAPSI (mNAPSI 75)
Results:
6 completed study to 36 weeks. Per-protocol analysis showed a reduction of mNAPSI by 64.1% (95% confidence interval, 46.5%-81.7%) from 33.8 to 12.3 at 36 weeks.
Mean percentage change in mNAPSI of the target nail (nail with highest baseline mNAPSI) at weeks 12, 24, 36, 48, and 52 compared with baseline decreased significantly at all time points, by between 50-60%, p<0.001.
mNAPSI75: Week 12 3/11 (27.2%)
Week 36 4/11 (36.4%)
Week 52 5/11 (45.5%)
Reason for discontinuation:
Gastrointestinal adverse events 3/6 (50.0%)
Lack of significant body psoriasis improvement 2/6 (33.3%)
Adverse events:
Drug related:
Nausea 5/11 (45.5)
Abdominal pain 2/11 (18.2)
Increased bowel movement 2/11 (18.2)
Possibly related:
Upper respiratory infections 2/11 (18.2)
Headache 1/11 (9.1)
Author Conclusions: Apremilast produced significant improvement in nail psoriasis, with results visible as soon as week 12.
Limitations: small sample size, high drop-out rate
Thoughts: spectrum of actual baseline disease severity not clearly outlined, other than baseline nail disease of mNAPSI>=5 – this isn’t high if the max score is 130. Hard to be sure how severe the baseline nail disease was
If patients enrolled were not on topical or any other therapy, one would assume baseline psoriasis was not that severe. Why were those on topical therapy not included? Topical therapy shouldn’t make much difference to nail disease.
Very high rate of GI side effects, in keeping with previous studies
Review:
Isolated nail lichen planus: An expert consensus on treatment of the classical form
Matilde Iorizzo, Antonella Tosti, Michela Starace, Adam I. Rubin, Martin Zaiac, Bianca Maria Piraccini, MD. Volume 83, ISSUE 6, P1717-1723
What: Literature review with aim of providing practical management suggestions for classical nail lichen planus (NLP), especially when disease is restricted to nails
Why: There are no guidelines for the management of NLP, which can lead to permanent destruction with functional and psycho-social consequences
Methods: Pubmed literature search. 21 papers identified
+ clinician survey to define: severity of clinical presentation; when systemic indicated/preferred; number of months necessary to evaluate the first results of treatment and the number of months over which a treatment is judged unsuccessful; and percentage of success of a treatment
Inclusion: cases of isolated NLP but also those where LP occurred in nails + other body areas due to limited literature available
Exclusion: not stated
Results/author recommendations:
Treatment of NLP is challenging with relapses being common
Treatment should be started early as NLP is aggressive
Lack of prospective studies with long-term follow up
It is not possible based on current data to predict who will and won’t respond to treatment
Fingernails may respond better than toenails due to faster growth rates
Topical treatments not recommended due to limited drug penetration and potential for side effects with long term use. Hydroxycloroquine and Methotrexate are not supported due to lack of evidence of efficacy
Treatment options – 1st line: IL/IM Triamcinolone
2nd line: PO retinoids
3rd line: azathioprine/cyclosporine/mycophenolate mofetil
Thoughts: it is interesting that in psoriatic nail disease, methotrexate is at least partially effective and acitretin makes little difference, whereas the authors have concluded the opposite for NLP. Does anyone have experience with treating widespread LP (skin and/or nails) with Methotrexate and was it of use?
BJD:
Translational Research
Frontal fibrosing alopecia shows robust T helper 1 and Janus kinase 3 skewing
E. Del Duca J. Ruano Ruiz A.B. Pavel R.D. Sanyal T. Song J. Gay‐Mimbrera N. Zhang Y.D. Estrada X. Peng Y. Renert‐Yuval R.G. Phelps R. Paus J.G. Krueger E. Guttman‐Yassky. Br. J. Dermatol., 183: 1083-1093. https://doi.org/10.1111/bjd.19040
What: Non-interventional, cross-sectional study, evaluating gene and protein expression in scalp biopsies of patient with frontal fibrosing alopecia (FFA), alopecia areata (AA) and healthy controls
Why: FFA is a progressive scarring alopecia, with unclear pathogenesis/molecular triggers/immune polarity and limited treatment efficacy. There is a lack of targeted treatments for FFA. By comparison, the cytokine profile of AA is better described, leading to targeted therapies in trial, such as JAK inhibitors. Comparison with AA may help elucidate pathogenesis of FFA
Methods:
Participants: 28 patients enrolled; 12 with FFA; 8 with mod-severe (>=30% scalp involvement) AA; 8 healthy controls
Biopsies: frontal scalp for FFA; leading edge in AA; non-lesional from >= 5cm from active areas
Severity evaluation: FFA severity index (FFASI) for FFA; SALT for AA
Spectrum: Mean FFASI for FFA 44.3 (range21–64) and mean SALT for AA 55.1 (range30.8–70.1)
Primary outcome: detection of CXCL9, used as biomarker of TH1. Two-sided Student’s t-test used for comparisons - paired for lesional vs nonlesional scalp and unpaired for FFA vs. controls, AA vs. controls and FFA vs. AA
Samples analysed for comparisons of:
- Histopathology and stem cell markers, with IHC to detect K15+ and CD200+, as markers of regenerative capacity of hair follicles
- Immune cell infiltration with IHC
- Immune and hair keratins using real time PCR (RT-PCR), to detect mRNA expression of preselected genes. Adjusted p values used for false detection rate
Results:
Histo/stem cell markers:
- Histo- in keeping with classic features of FFA and AA
- Stem cell markers: K15+ cells were diminished in FFA lesional scalp cf AA and controls (p<0.05).
CD200+ cells at bulge level were reduced in FFA and AA scalp cf controls, achieving significance for FFA lesional scalp (P < 0.001). Significant reduction in CD200+ cells in lesional FFA cf nonlesional scalp
Immune cell infiltration:
- Significant increase in CD8+ T cells in lesional FFA and lesional AA (P<0.05)
- FOXP3 Treg significantly increased in FFA lesional scalp cf healthy scalp (p<0.01) and lesional AA (P<0.05)
- Prominent co-localisation of CD3+/CD8+ infiltrate in the bulge of lesional FFA and the bulb of lesional AA
Immune and hair keratins:
- Markers of T‐cell/NK‐cell activation (IL‐2, IL‐15, IL‐2RA, IL‐15RA, IL‐16), cytotoxic T cells/NK cells (granzyme B), and general inflammation (PDE4A, PDE4B) were significantly increased in FFA cf AA in both lesional and nonlesional tissues – p<0.05
- Expressions of Th1 markers (IFN‐γ, chemokine ligand (CCL)5, CXCL9, CXCL10, IL‐12RB1, STAT1) were significantly increased in FFA scalp cf controls, with lesional FFA expression significantly elevated compared with FFA nonlesional tissues and AA lesional tissues – p<0.05
- JAK3 was significantly induced in FFA lesions (P < 0·001), while JAK1 was not upregulated in either FFA or AA
- Fibrosis markers were significantly elevated in FFA cf AA lesional tissues [transforming growth factor (TGF)β1, plasminogen activator inhibitor (PAI)1, VIM, fibronectin (FN)1, collagen (COL)1A, COL3A, SNAIL2] and in FFA cf controls (PAI1, VIM, FN1, connective tissue growth factor, COL1A, COL3A) – p<0.05
- FOXP3 significantly elevated in FFA cf both AA lesions and controls – p<0.05
- All measured hair keratins (KRT84/85/75AP1-1/35/40/83) were significantly downregulated in AA lesional scalp vs. nonlesional or normal scalp, while most were not reduced in FFA
- FOXP3 mRNA expression was the best single‐gene classifier, discriminating FFA and AA lesions with almost 100% accuracy (area under the curve 0·96)
Author Conclusions: T helper 1/JAK3 mediated follicular damage and fibrosis may result in FFA
CD200 and K15+ loss at the bulge in FFA is juxtaposed with CD8+ infiltration
The even greater Th1 overexpression in FFA vs. AA tissues, and significant upregulation of JAK3 and STAT1, in FFA compared with healthy control scalp and even AA scalp, possibly suggest a therapeutic potential for JAK inhibitors in halting disease progression in early FFA
Hair keratins are not downregulated in FFA, unlike in AA.
Thoughts:
Would be of interest to know how JAK2 induction if affected in FFA vs AA as this could widen therapeutic options to include baricitinib
Qualitive and Outcomes Research
Development of the Scalp Hair Assessment PRO™ measure for alopecia areata
K.W. Wyrwich H. Kitchen S. Knight N.V.J. Aldhouse J. Macey F.P. Nunes Y. Dutronc N. Mesinkovska J.M. Ko B.A. King
What: cross‐sectional, noninterventional, qualitative interview study
Why: to create a content-valid and clinically meaningful patient reported outcome (PRO) for alopecia areata (AA), with scores comparable with the five‐response‐level Alopecia Areata Investigator Global Assessment (AA‐IGA). PRO’s are important for evaluating treatment efficacy.
Methods:
Expert interviews: 10 US clinical experts in AA were underwent qualitive interviews to develop a draft PRO
Patient interviews: the draft PRO content-validity was tested through 2 rounds of semi-structured interviews, with patients with AA who had experienced ≥ 50% scalp hair loss. 1st: open-ended questions to explore patient experiences of AA signs and symptoms and amount of improvement they would consider meaningful. 2nd: cognitive debriefing explored patients’ acceptance, opinions and interpretation of the draft Scalp Hair Assessment PRO. Performed by qualitive interviewer trained in PRO development techniques
Patient Inclusion: aged ≥12 years at time of consent (Round 2 aged 18-60 years); severe AA diagnosed by a clinician and determined by SALT ≥50%; a history of AA episodes lasting >6 months but <8 years; successful JAKi treatment but SALT ≥50 prior to treatment (Round 1 only); recruited from centres in North America. Round 1: n=30 / Round 2: n=15
Exclusion: taking part in round 1; other forms of alopecia; any severe dermatological condition or other severe medical condition which could impact the study
Conceptual saturation analysis used to determine a sample size of 30 as ample for round 1
Demographic: R1, M:F = 13:17. R2, M:F = 6:9. Mean age- R1 = 35·2 ± 16·9 and in R2 = 29·5 ± 8·4
Spectrum: all >50% scalp hair loss and 24/30 had also partial/full eyebrow and or eyelash loss. Average salt scores: R1 = 57·9 ± 40·4 and R2 = 85·9 ± 20·3. JAKi experienced: R1 = 18 (60) and R2 =2 (13)
Results:
Patient interviews: Scalp hair loss was the most bothersome symptom for most patients (n = 35, 78%). Patients described feeling self‐conscious or insecure (n = 25), feeling embarrassed or ashamed (n = 10) and lack of confidence (n = 9). Patients deemed treatment success at a median 80% in round 1 (range 30–99%; n = 26) and median 75% in round 2 (range 50–100%; n = 15). Most patients considered a treatment successful even if the newly grown hair was different in colour, quality or thickness from their original hair (round 1, 15 of 20; round 2, 13 of 15)
The term ‘hair loss’ was frequently misinterpreted by patients in round 1, resulting in this being rephrased to ‘missing hair’ in round 2
In round 2, 12/15 agreed with the following PRO, modified from round 1:
No missing hair (0%); limited area (1-20%); moderate area (21-49%); large area (50-94%); nearly all or all (95-100%)
Author conclusions: There was agreement among patients that successful treatment would result in ≤ 20% hair loss
While the Scalp Hair Assessment PRO was developed to inform clinical trial endpoints, this measure has potential utility in clinical practice to understand hair loss status prior to treatment, to facilitate discussion between patients and clinicians, and to achieve a shared understanding of treatment goals.
Thoughts: most of our patients in UK are not yet treated with JAK-I for AA, so does their inclusion criteria result in this scale being content-valid for a UK population?
Seems strange to use separate patients between rounds. Surely better to gain opinions of the PRO from the people whom you have spent the time speaking to and exploring their experiences of AA?
‘Caucasians’ heavily over-represented in round 1. ‘Black or African American’ under-represented in rounds 1 and 2
Symptoms are not included in this PRO
Research Letter
Frontal fibrosing alopecia: a descriptive cross‐sectional study of 711 cases in female patients from the UK
S.M. McSweeney E.A.A. Christou N. Dand A. Boalch S. Holmes M. Harries I. Palamaras F. Cunningham G. Parkins M. Kaur P. Farrant A. McDonagh A. Messenger J. Jones V. Jolliffe I. Ali M. Ardern‐Jones C. Mitchell N. Burrows R. Atkar C. Banfield A. Alexandroff C. Champagne H.L. Cooper G.K. Patel A. Macbeth M. Page A. Bryden M. Mowbray S. Wahie K. Armstrong N. Cooke M. Goodfield I. Man D. de Berker G. Dunnil A. Takwale A. Rao T.‐W. Siah R. Sinclair M.S. Wade K. Bhargava D.A. Fenton J.A. McGrath C. Tziotzios
What: descriptive cross‐sectional study of the clinical phenotype in women from the FFA UK genome‐wide association study (GWAS) cohort
Methods: from the GWAS cohort, selected patients were assessed for multiple clinical variables based on a standardized pro forma. Descriptive and exploratory statistical analysis then undertaken
Inclusion: female patients with a formal diagnosis of FFA made by a consultant dermatologist from 20 secondary care dermatology departments across the UK; Eurasian ancestry;
Exclusion: participants with missing data
Results: Phenotypic data were available for 711 UK women with FFA among a total of 1044 participants in the GWAS cohort
Median age = 66 (interquartile range 59–72); median duration of scalp hair loss = 7 years (interquartile range 5–10); of 485/663 (73·2%) participants with available data, frontotemporal hairline recession occurred following menopause.
Clinical features n (%): Perifollicular erythema 508 (77·3); Follicular hyperkeratosis 119 (26·0); Occipital recession 178 (26·0); Eyebrow loss 620 (90·6); Eyelash volume loss 311 (44·5); Limb hair loss 543 (77·5); Axillary and pubic hair loss 464 (67·0); Nail changes (any type) 165 (23·7)
PMHx: Autoimmune disease 141 (20·7); Autoimmune thyroid disease 88 (12·9); Previous oestrogen deficiency 38 (5·6)
Treatments: 44% prescribed a medication for their FFA; hydroxychloroquine in 24·0%; topical corticosteroids in 16·6%; oral tetracycline antibiotics in 10·1%; topical calcineurin inhibitors in 3·8%; intralesional steroids in 1·7%; oral corticosteroids in 1·3%; and immunosuppression/retinoids in 2.1%
Author conclusions: prevalence of AI disease/oestrogen deficiency more prevalent than general population.
Thoughts: surprising that so few were offered treatment. Perhaps clinician attitudes regarding the efficacy of available treatments and indeed limited efficacy in practice has caused this. Would be interesting to know how many were offered wigs.